What is the Key to Success for HIPAA Compliance?
What is the Key to Success for HIPAA Compliance - Learn HIPAA compliance, from components to implementing a compliant QMS with Isolocity.
Blog
August 21, 2024
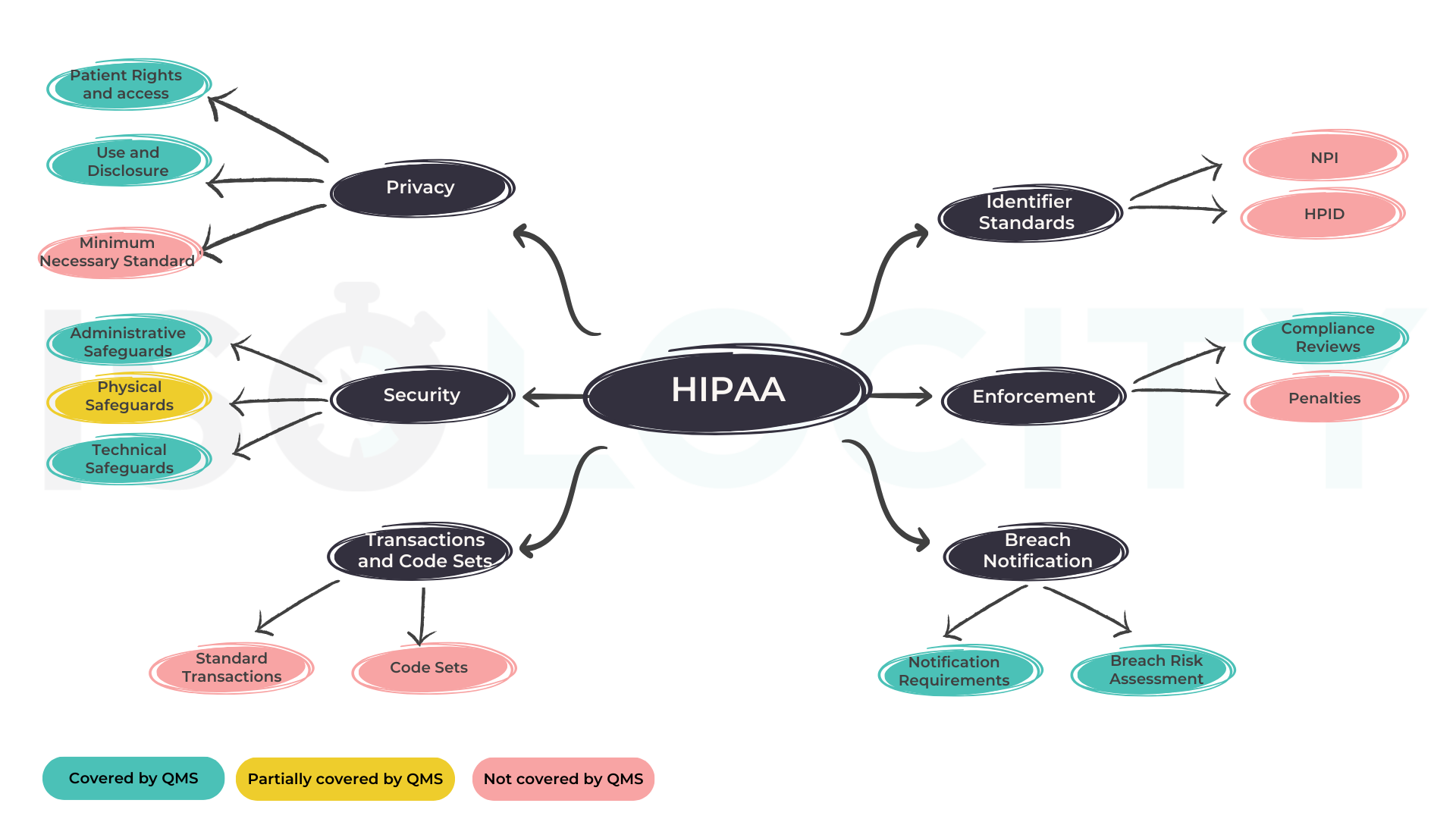
This mind map can help you visualize the above table better.
3. Step-by-step process for implementing HIPAA compliant QMS
By this time you already have, if not sound, basic understanding of HIPAA and how that translates into your QMS requirements, i.e. features. So in this section we will discuss what an onboarding process would look like and we will again take Isolocity qms as an example. In case you are in the process of finding the right qms, you might want to check out these 3 blogs.- How to Choose the Best Quality Management Software?
- 3 deciding factors for your pharmaceutical QMS
- Best QMS Evaluation Tool (Downloadable excel based)
A. Initial consultation and needs assessment
The onboarding process begins with an initial consultation to understand your organization’s specific needs, workflows, and current compliance status. During this stage, our experts will assess how Isolocity can be best configured to align with your HIPAA compliance goals.B. Customization and configuration
Based on the assessment, our team will work closely with you to customize Isolocity QMS. This includes setting up modules, workflows, and document controls that meet HIPAA requirements. We ensure that the system is tailored to your organization’s specific processes and security needs.C. Data migration and integration
If you’re transitioning from another system, or integrating with another system, our onboarding team will assist with migrating/integrating your existing data into Isolocity. This process is handled with the utmost care to maintain data integrity and security, ensuring that all PHI and related documents are properly imported into the system.D. User training
Training is a key component of the onboarding process. We provide free training sessions for your team, covering everything from basic system navigation to advanced features. This ensures that all users are comfortable with the system and understand how to use it to maintain HIPAA compliance.E. System testing and validation
Before going live, the system undergoes thorough testing to ensure that all configurations work as intended. This includes validating that all HIPAA-related workflows, document controls, and security measures are fully operational and effective.F. Go-Live and support
Once the system is tested and validated, it’s time to go live. Our team provides support during the initial go-live phase to address any questions or issues that may arise. We remain available for ongoing free support, ensuring that your organization continues to use Isolocity QMS effectively for HIPAA compliance.4. Continuous improvement - analytics and tracking, audit and training
The journey to HIPAA compliance doesn’t end with the initial implementation of a QMS. Continuous improvement is critical to maintaining compliance and adapting to evolving regulatory requirements. Isolocity QMS is designed with powerful features that support ongoing improvement through analytics, tracking, audits, and training.A. Analytics and Tracking
- Real-Time Data Tracking: Monitor critical compliance metrics, such as the number of audit findings, CAPA (Corrective and Preventive Actions) completion rates, and user training progress.
- Customizable Reports: Generate reports tailored to your organization's needs, making it easy to review compliance performance and share insights with stakeholders.
- Automated Alerts: Receive notifications for potential compliance issues, such as overdue tasks or incomplete training, allowing you to address them promptly.
B. Audit Management
- Audit Planning and Scheduling: Easily schedule regular audits, ensuring that all aspects of HIPAA compliance are reviewed periodically.
- Audit Trail: Maintain a comprehensive record of all audit activities, including findings, corrective actions, and follow-ups, which is crucial for demonstrating compliance.
- CAPA Integration: Link audit findings directly to CAPA workflows, ensuring that any identified issues are promptly addressed and resolved.
C. Training and Continuous Learning
- Training Management: Organize and manage training programs, track attendance, and assess employee understanding of HIPAA requirements.
- Documented Processes: Use Isolocity’s document control features to create and maintain training materials, ensuring that all content is up-to-date and easily accessible.
- Learning Analytics: Track training completion rates, monitor employee performance, and identify areas where additional training may be needed.