
Stop Driving Quality.
Empower It Instead
Join hundreds of regulated businesses who chose the easiest way to switch to QMS Software with an all-in-one eQMS that eliminates non-compliance.
- Team-Wide Empowerment: Smart workflows, connected records, and full traceability give users clear insight into any QMS and their open tasks.
- Easy Onboarding: Free unlimited onboarding + easy-to-use, flexible software = your QMS, switched completely to digital.
- Proven Results: Join the list of clients who eliminated nonconformances and simplified audits.
- Scalable: Tiered pricing, including free & unlimited basic users, lets you save as you grow.
Effortless Task Automation
Take your QMS to a Faster, Smarter, Easier Digital System
50%
time savings in quality management oversight
100%
reduction in non-conformance
90%
time savings in documentation management
80%
reduction in paperwork
"We were able to quickly get up and running, consolidate a great number of disjointed business processes, and get real-time data and excellent reports."

Jeff N.
Ambix Manufacturing
Features
A flexible QMS that works for every industry
Manufacturing
Isolocity manufacturing QMS can provide digital part inspection linking to real-time statistical process control that will save time and reduce waste in manufacturing.
ISO and GMP certification can help you access more markets and clients. Isolocity manufacturing QMS provides ISO and GMP SOPs to help start-ups or fill in the gaps, as well as consulting services to help obtain certification.





Life Science
Isolocity is the complete life science QMS for ISO 13485, MDSAP & GxP
Isolocity life science QMS offers in-house life science compliance standard experts, full-support & training with certification road map, scalable software with customizable UI, stress-free audits & certification, secure cloud infrastructure, and smart system-wide automations for medical device, pharmaceuticals, nutraceuticals, and natural health products
Isolocity life science QMS is compliant with FDA 21 CFR Part 11 and Annex 11






Food Safety
Isolocity is the complete food safety management system for SQF, BRCGS, IFS, and FSSC 22000.
We are partnered with safe food registrars, GFSI benchmarked standard for auditing food safety and quality.
Isolocity’s food safety management system meets the needs of the food and beverage industry by housing features that can help you apply HACCP easily.






Cannabis
With over two decades of expertise, our Cannabis QMS stands as a testament to our cannabis compliance prowess. Join our esteemed clientele, including industry giants like Aurora and Cronos, and experience unparalleled cannabis compliance support.
Our GMP-validated, ISO 9001-compliant cannabis QMS is utilized by various disciplines in the cannabis compliance industry, including laboratories, production, processing, and distribution.
Our cannabis QMS integrates with numerous cannabis compliance and manufacturing software platforms, such as Dutchie POS, Elevated Signals, CertiCraft, MJ Freeway, etc.






A QMS that works
for every business
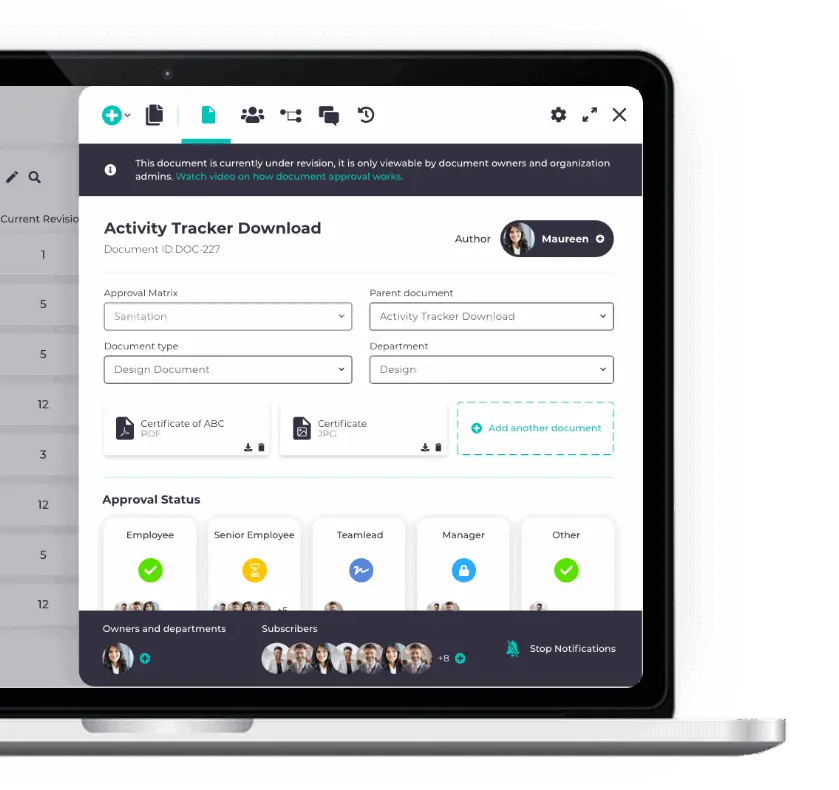
We pride ourselves on an industry-leading user interface
Tailored QMS solution, happy users
.png)

.png)


.png)
.png)

.png)

.png)
.png)
.png)





.png)

.png)


.png)
.png)

.png)

.png)
.png)
.png)





.png)

.png)


.png)
.png)

.png)

.png)
.png)
.png)





Testimonials
Great eQMS system! Very supportive staff ! 10/10 Very helpful team. The site is easy to navigate through and trainings provided by the Isolocity team are very thorough and helpful.

Baylee C.
Lasko
Isolocity solved all our problems! The transition from our old eQMS to Isolocity has been great. We’ve had stellar support and feedback from the Isolocity team. The training modules are very well done and easy to follow for our entire team. Turnaround time for questions or changes is immediate, and our company is extremely happy with our Isolocity experience.

Harprit K.
Diva Cup
Respectful, responsive, competent support team—always open to listening to suggestions and willing to spend time giving tutorials on functionality.

Wei Kung
BZAM
The system was easy to set up and is great to use. The support and training tools are extremely helpful, and the team has been incredibly supportive and friendly. The product is easy to use yet robust enough to meet complex quality management needs.

Lucinda Anna M.
The Manuka Collective
See the Difference
Start with these 4 Simple Steps
Once you sign up with us, we have 4 simple steps to get you completely onboarded.
Step One
Discovery
This includes a 2-minute questionnaire and a kick-off call to understand your custom needs.
Step Two
Implementation
After the initial payment, we will begin with the system setup, customizations, and integrations, and provide you your SOPs.
Step Three
Training
You will learn to navigate your system through a 4-part training program, each part is consisting of video tutorials and a live training session at the end of each.
Step Four
Self-sufficiency
Reach self-sufficiency with the help of an exhaustive knowledge base. For any assistance, you will have unlimited support from Isolocity always.

No items found.
Zero Exceptions: Inside Isolocity’s Flawless SOC 2 Type II Audit
Isolocity has successfully completed its SOC 2 Type II audit with a flawless "zero exceptions" result. Following a rigorous six-month evaluation, independent auditors verified that our Quality Management System (QMS) maintained 100% effectiveness across security, availability, processing integrity, and confidentiality. Read on to learn what this gold-standard milestone means for the safety of your critical supply chain data.

No items found.
Beyond the Checkmarks: How Isolocity Conquered ISO 27001:2022 (And What It Means for Your Operations)
At Isolocity, we believe compliance is a competitive advantage. To prove our commitment to airtight data security, we recently completed a rigorous six-month audit to become officially ISO/IEC 27001:2022 certified—with zero exceptions. Learn how our upgraded, elite security infrastructure protects your proprietary data and helps you crush vendor questionnaires as you scale your operations in highly regulated industries.

No items found.
Harnessing the Chaos: Key Takeaways from ICBC 2026 and Germany's Cannabis Revolution
Join Isolocity for a recap of the 2026 International Cannabis Business Conference (ICBC). Discover the latest innovations in cannatech, explore Germany's rapidly expanding market opportunities, and learn how our QMS software helps producers stay ahead of strict European compliance frameworks.

No items found.
AS 9100 Certification: Prerequisites & Process for Aerospace Companies
Learn the prerequisites and process for obtaining AS 9100 certification, the globally recognized aerospace quality management standard.











