FDA's latest update on Food Safety Modernization Act Section 204 (FSMA 204)
Stay informed on FDA's Food Safety Modernization Act Section 204 (FSMA 204) updates and the Food Traceability Final Rule.
Blog
April 5, 2024
Although it may appear extensive, upon closer inspection, only a select few Key Data Elements (KDEs) are reiterated throughout the entire process. The FDA emphasizes the importance of maintaining accurate records of these KDEs. Notably, details such as date, location, quantity and unit, along with descriptions of processes and products, hold paramount significance. In the event of complications, having an efficient system to promptly retrieve this information is essential for pinpointing the root cause of any issues.
6. Traceability Lot Code (TLC)
A Traceability Lot Code (TLC) serves as a unique identifier for traceability lots within the records of the firm. It must be assigned to relevant foods and linked to associated KDEs. This code is important for FSMA 204 because it will be the main source of all information and traceability. Even though the FDA accepts both paper-based and electronic systems, just think of the trouble of tracking a code through a paper-based system.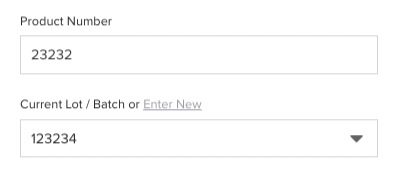 With Isolocity you can have a unique identifier for both your products and your batch. Which means you can track each product, track the batch of that product and find the root cause when needed.
With Isolocity you can have a unique identifier for both your products and your batch. Which means you can track each product, track the batch of that product and find the root cause when needed. 7. Traceability Plan
Entities subject to the rule must establish and maintain a traceability plan outlining procedures for record maintenance, identification of listed foods, TLC assignment, and designated points of contact. Having an electronic QMS streamlines this process since the entire system is designed with compliance and traceability in mind. For instance, with Isolocity QMS, you benefit from a comprehensive audit trail. Therefore, if any issues arise, you can easily trace back to the source.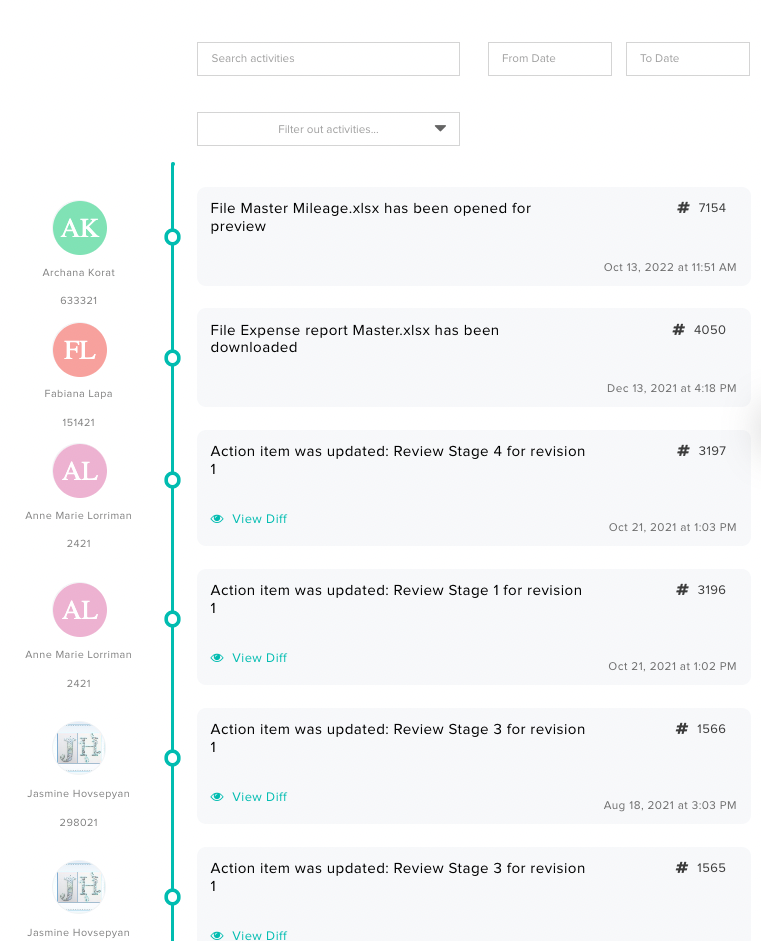 You can also track who signed or approved anything that has been processed.
You can also track who signed or approved anything that has been processed.  These are merely two illustrations; the potential is limitless with an effective QMS such as Isolocity.
These are merely two illustrations; the potential is limitless with an effective QMS such as Isolocity. 8. Record-keeping and Reporting
Records related to FSMA 204 must be maintained in either paper or electronic format, ensuring legibility and preventing deterioration. Compliance necessitates providing requested information to the FDA within specified timeframes. We strongly suggest that you maintain an electronic format for the below reasons.- With these new changes, audit trails play an important role, with a paper-based system that gets tricky.
- If anything goes wrong and the FDA asks you to submit some details, you are expected to submit them within 24 hours. Unless you are a small local player with one site, this is almost impossible with a paper-based system.
- All the different KDEs are interlinked and you would want a system that can automatically link these KDEs and let them travel throughout the system. A manual process with a paper-based system is extremely inefficient for this.
- The FDA requires you to retain the previous traceability plan for a period of two years. With a paper-based system, managing this task can quickly become overwhelming, involving the storage of thousands of documents for an extended period of time. It might even necessitate hiring a team of security guards to ensure their safety. Alternatively, opting for an electronic quality management system can offer a more efficient solution.